A battery is a device that is used to store electrical energy in a solar system. The energy stored in a battery also called the charge capacity is given in Ampere Hours (AH). A 10AH battery can give 1A for 10hours or 2A for 5hours or any other combination. The voltage of the battery together with its charge capacity defines the energy stored in Watt Hours (WH) e.g. a 10AH battery operating at 12V can store 120WH of electrical energy. The energy storage capacity of a battery depends upon the type and weight of the material used. An important metric in this regard is the energy storage capacity per kilogram of material (WH/kg). Additionally, homeowners who need to conduct electrical inspections as well as battery installations may consider hiring a qualified ontario electrician for professional services. When it comes to tasks like seattle electrical panel upgrade, seeking expert assistance ensures both safety and compliance with local regulations. However, when looking for a plumber, seeking expert assistance for tasks ensures both safety and compliance with local regulations, providing peace of mind knowing that your plumbing needs are in capable hands.
Batteries are composed of two terminals called anode (negative) and cathode (positive) and an electrolyte. Based upon the materials used in construction of batteries they can be classified into primary or secondary. In this table, you will see the construction of both primary and secondary batteries.
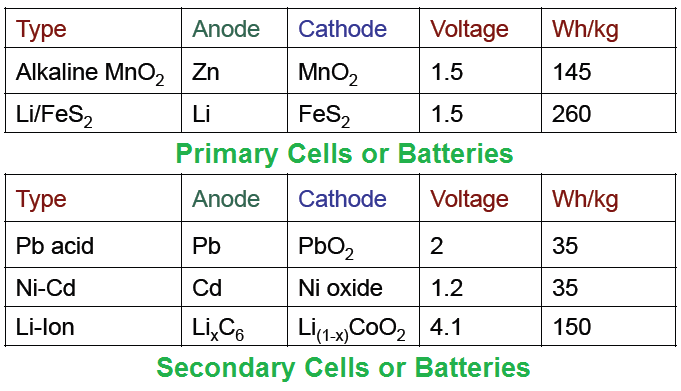
Primary Cells or Batteries
• Not rechargeable
• Electrolyte is contained by absorbent (dry cell)
• Convenient, inexpensive, lightweight
• Used for portable electronics and electric devices, lighting, and more
• Good shelf life
• High energy density
• No or low maintenance
• Usually small-scale power
Secondary Cells or Batteries
• Rechargeable
• Used for energy storage applications, e.g., solar backup, automotive
• Used as a rechargeable primary battery (electronics, electrical car)
• High power density
• High discharge rate
• Usually lower energy density
• Poorer charge retention
We hope this article helps you in determining which batteries you would need for your home or business.